|
5/1/2023 0 Comments Isoelectric point measure 
The titration conditions are defined in table 1. This temperature ensured that equilibration of sample viscosity was minimized as the bulk of the sample situated external to the Nano instrument was at a room ambient temperature of 20˚C.įigure 3 shows the zeta potential measured as a function of pH for a titanium dioxide sample dispersed in deionised water. The following examples were all measured on a Zetasizer Nano ZS instrument in conjunction with an MPT2 at 20˚C. The measurement of particle size, zeta potential or the intensity of scattered light can be programmed depending upon which Zetasizer Nano instrument is used. If the change in conductivity wanted to be studied, an appropriate salt would be used as a titrant in a logarithmic additive titration.Īfter the conditions have been changed, the sample is then circulated into the disposable capillary cell situated in the Zetasizer Nano for the measurements to be made. For example, if the effect of changing the pH on the zeta potential of a sample wanted to be studied, the titrants would be acid and base. The titrants used will depend upon the type of titration requested. A typical titration schematic showing the ability to have three titrants for changing the composition of the sample and circulating it into the disposable capillary cell situated in the Zetasizer Nano. The MPT2 has the ability for three titrants to be used to change the composition of the sample and can be programmed to perform the following titration types:įigure 2. 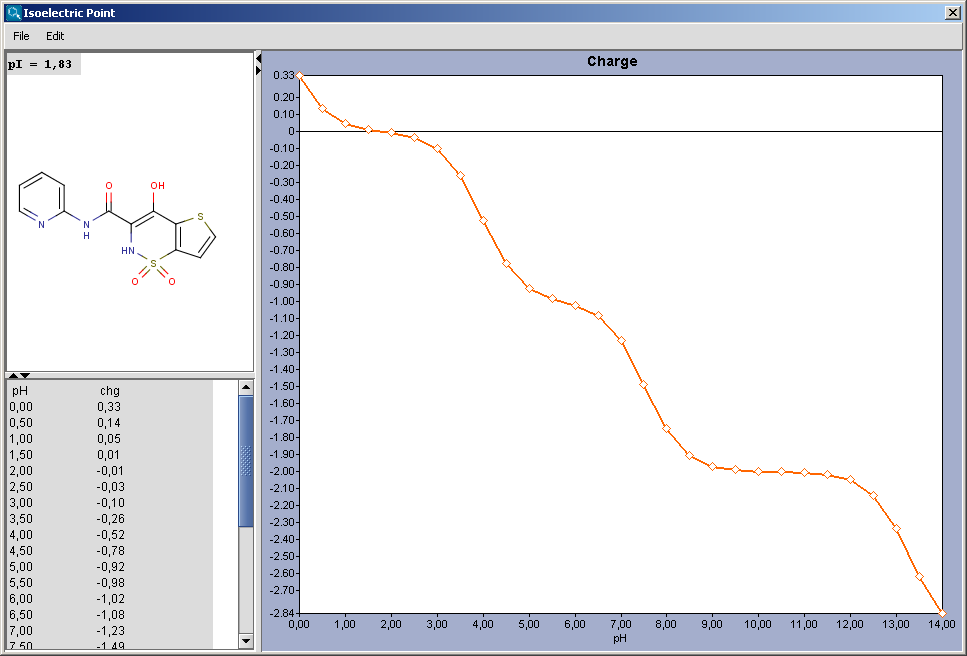
The MPT-2 is an integrated system designed to automate changes in the composition of the sample and then transfer the sample to the optics unit for measurement of the size, zeta potential and intensity.įigure 2 shows a typical titration schematic for the MPT2 linked to a Zetasizer Nano. However, automatic determination is less time consuming and more desirable and can be achieved by combining a Zetasizer Nano instrument with a Multi Purpose Titrator MPT2. This procedure can be carried out manually. In order to determine whether a sample has an IEP or not, the conditions of the sample need to be altered and the effect on the zeta potential monitored. At these extremes of pH, the forces resulting from this electrostatic repulsion should be sufficient for the sample to resist flocculation. In addition, at pH values greater than 8, there is significant negative charge present. A plot of the zeta potential of a sample measured as a function of pH.įor example, at pH values less than 4, there is significant positive charge present. In addition, the plot can be used to predict that the sample should be stable at the extremes of pH.įigure 1. In this example, the isoelectric point of the sample is at approximately pH 5.5. This may be important when considering the shelf life of a product, as normally the sample needs to be away from the IEP.įigure 1 shows a typical plot of the zeta potential of a sample measured as a function of pH. For a sample which is electrostatically stabilized, the IEP is often the point of least stability due to the repulsive forces being weakest. The isoelectric point (IEP) is defined as the point of zero zeta potential. Further information on this technique can be found in the technical note “Measuring Zeta Potential: Laser Doppler Electrophoresis” (KB000606) available on the Malvern Panalytical web site. The measurement of zeta potential can be performed on a suitable Zetasizer Nano instrument using the technique of laser Doppler electrophoresis. The effect of the pH, or ionic strength of the medium or the concentration of an additive on the zeta potential can give information in formulating the product to give maximum stability.įurther information on zeta potential can be found in the technical note “Zeta Potential: An Introduction in 30 Minutes” (KB000734) available on the Malvern Panalytical web site. Zeta potential is influenced by pH, ionic strength (the concentration and type of ions present) and the concentration of any charged molecules in the dispersant. 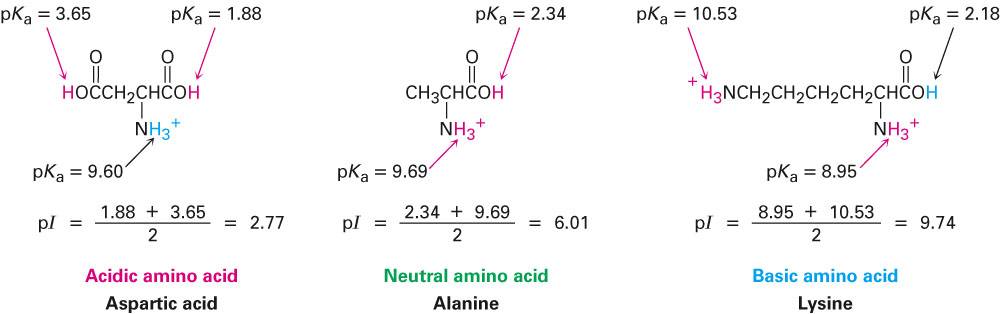
However, if the particles have low zeta potential values then there is no force to prevent the particles coming together and flocculating. If all the particles in suspension have a large negative or positive zeta potential then they will tend to repel each other and there is no tendency for the particles to come together. The Zeta Potential of a colloidal dispersion is a measure of the electrostatic repulsion between particles and can be used as an indicator of dispersion stability. Sponsored by Malvern Panalytical Oct 24 2007 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
